by Vanessa Göschl
Photos by: Vanessa Göschl

Vanessa Göschl
PhD student | Molecular Signal TransductionYou really have to like what you’re doing. And with this, I don’t mean that you’ll have to like every single task of every day – because this for sure isn’t possible. But you need to have something that grounds and motivates you, when times get stressful and experiments don’t work out.
The fact that I still like my project just as much as I did in the beginning, is a great motivation to keep on going. But sometimes, when I need an outlet for stressful times, I enjoy doing basic cell culture work, such as medium changes, where I can calmly focus on a ‘simple task’, while rearranging my thoughts and clearing my head. If this doesn’t help, go home, get some rest and start again fresh tomorrow!
I think there are a lot of positive aspects to doing a PhD. Since the beginning of my bachelor’s studies, I knew that I loved working with cells, planning and doing experiments and diving deeper and deeper into a specific research topic. This hasn’t changed – and now, I can do exactly this!
One of the many perks of doing a PhD is definitely the chance to design and perform your own experiments and to be able to plan your working hours around the experiments if necessary (which of course can be a pro and a con for the work-life-sleep balance). However, I really enjoy not having a strict daily routine but rather doing different tasks. At the Institute I work in, we have a great community of PhD students and also supervisors, facilitating the exchange of knowledge, pitfalls, trouble shootings and room for improvement, which is highly motivating.
I would say: wander around as much as you can. Go by foot through Vienna and look around – there are plenty of buildings with statues and beautiful ornaments on the walls, artistically decorated windows and balconies. Although I was born and raised in Vienna, I don’t think I will ever get bored by this.
Hi! My name is Vanessa and I am a third year PhD student at the Medical University of Vienna. In my PhD-project I am trying to understand how toxic accumulations of glutamate can occur inside the brain upon different kinds of diseases or as a result of blunt force trauma to the head (for example, you accidently fall and hit your head).
Let´s discuss briefly, what is Glutamate?
Glutamate is an amino acid in our body that is synthesized in the brain and acts there as a signaling molecule, to help our neurons, or nerve cells, to communicate with one another. In other words, it acts as a neurotransmitter in the brain and as such, it plays a crucial role in regulating our nerve signals, energy production and metabolism. Having that said, Glutamate effects many functions in our body. It determines how well our senses are working and how we perceive pain, light and sound. It affects our ability to learn and remember, as it plays an important part in how our brain forms memory. It can also increase or decrease our energy levels and alertness, and regulates our sleep-wake cycle, which has a direct impact on our mood and wellbeing.
However, when glutamate is released in large quantities and the breakdown processes of the body are not fast enough, toxic build-ups can occur in the brain. These toxic build-ups can lead to overstimulation of nerve cells, resulting in cell damage and cell death. Glutamate mediated excitotoxicity is a common hallmark in many neurodegenerative diseases such as stroke, trauma, Alzheimer's, Parkinson's and epilepsy.
You might wonder “How does the breakdown process work?” Upon a stimulus, an action potential is formed, which leads to the release of glutamate, which can then activate receptors on other nerve cells, creating another action potential and thereby forming a signaling cascade. Although this mechanism is usually tightly regulated, like all systems, it can have its flaws or can be disrupted. To investigate how the dysregulation of glutamate signaling occurs, I am primarily using two different methods: Electrophysiology and Fluorescence microscopy. Electrophysiology: In the picture, you can see an electrophysiology setup, with a microscope in the center, a perfusion system to the left and to the right a micropipette with an electrode inside, that is ultimately connected to a computer to visualize the electrophysiological signal. During an experiment, the micropipette is closely attached to a cell to form a tight seal onto the cell membrane, making it possible to record intracellular activity, including mechanisms such as ion exchange and receptor activity or firing of action potentials. With the perfusion system, it is now possible to test how the cellular activity changes as a response to the application of different substances or stimuli, by placing the perfusion outlet in close distance to the cell of interest and gently flushing the surrounding environment with a liquid of choice. Since each reservoir of the perfusion system is connected to the perfusion outlet via an individual tube, multiple substances can be tested this way.
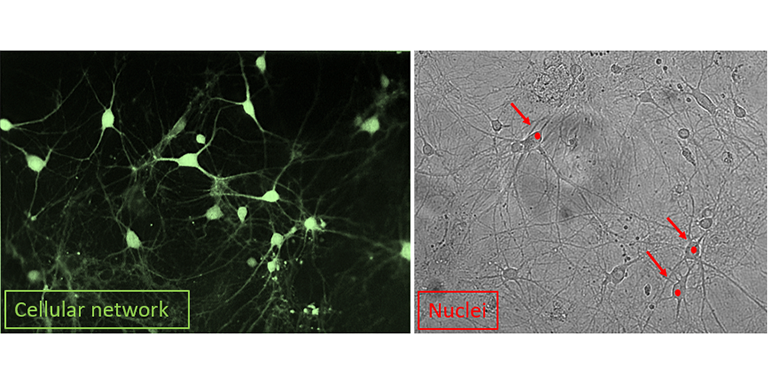
Fluorescence microscopy: In addition to electrophysiology, I am also interested in changes in the cell morphology (shape and size of the cell) and the overall survival of my cells. For this, I am using different dyes to visualize the area of interest, e.g. whole networks of cells or single nuclei from dying cells. With these approaches, it will be possible to identify some of the key players involved in this mechanism. Aside from the fact that any one of us could suffer a head injury at any moment, the number of people with Alzheimer's and Parkinson's disease continues to rise worldwide and there is a great need for answers from both patients and the public. I hope that my research will lead to a better understanding of this process and ultimately to the development of new treatments that are not only symptomatic but also preventive.
